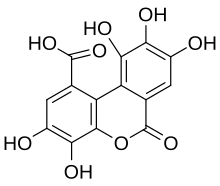
Luteic acid
Luteic acid
Names
Preferred IUPAC name
3,4,8,9,10-Pentahydroxy-6-oxo-6H -dibenzo[b ,d ]pyran-1-carboxylic acid
Other names
Luteolic acid
Identifiers
ChemSpider
UNII
InChI=1S/C14H8O9/c15-5-2-4-7(11(19)9(5)17)8-3(13(20)21)1-6(16)10(18)12(8)23-14(4)22/h1-2,15-19H,(H,20,21)
Key: FLZGFQFYDGHWLR-UHFFFAOYSA-N
InChI=1/C14H8O9/c15-5-2-4-7(11(19)9(5)17)8-3(13(20)21)1-6(16)10(18)12(8)23-14(4)22/h1-2,15-19H,(H,20,21)
Key: FLZGFQFYDGHWLR-UHFFFAOYAI
C1=C2C(=C(C(=C1O)O)O)C3=C(C(=C(C=C3C(=O)O)O)O)OC2=O
Properties
C14 H8 O9
Molar mass
320.21 g/mol
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
Luteic acid is a natural phenol found in numerous fruits . It is a monolactonized tergalloyl group. Maximilian Nierenstein showed in 1945 that luteic acid was a molecule present in the myrobalanitannin, a tannin found in the fruit of Terminalia chebula ellagic acid .[1] hexahydroxydiphenic acid . It is also present in the structure of the tannins alnusiin and bicornin .[2]
References
^ Nierenstein, M.; Potter, J. (1945). "The distribution of myrobalanitannin" . The Biochemical Journal . 39 (5): 390–392. doi :10.1042/bj0390390 . PMC 1258254 PMID 16747927 . ^ Structures of alnusiin and bicornin, new hydrolyzable tannins having a monolactonized tergalloyl group. Yoshida T, Yazaki K, Memon M.U, Maruyama I, Kurokawa K, Shingu T and Okuda T, Chemical and pharmaceutical bulletin, 1989, volume 37, number 10, pages 2655-2660, INIST 19467830 (abstract )
Moieties Lactones Monomers
Acetonyl geraniin
Alnusiin Bicornin Carlesiin
Casuarictin Emblicanin A and B
Euscaphinin
Galloyl pedunculagin
Grandinin Helioscopinin B
Jolkinin
Lagerstannin A, B and C
Macranganin
Myrobalanitannin
Nupharin A , B, C, D, E and FPedunculagin Punicalagin Punigluconin Phyllanemblinin A, B, C, D, E and F
Punicalin Roburin E
Rugosin E
Sanguiin H-5
Stenophyllanin A , B and CStrictinin Tellimagrandin I and II Teracatain
Terchebulin
Terflavin A and B
Tergallic acid Tergallic acid dilactone C-glycosidic ellagitannins Dehydroellagitannins (molecules withdehydrohexahydroxydiphenic acid (DHHDP) Transformed ellagitannins
molecules with chebulic acid molecules with Elaeocarpusinic acid
Elaeocarpusin
Helioscopin B
Mallojaponin (1-O-Galloyl-2,4-elaeocarpusinoyl-3,6-(R)-valoneayl-beta-D-glucose)
Oligomers Other
The article is a derivative under the Creative Commons Attribution-ShareAlike License .
A link to the original article can be found here and attribution parties here
By using this site, you agree to the Terms of Use . Gpedia ® is a registered trademark of the Cyberajah Pty Ltd