Patuletin
Patuletin
Names
IUPAC name
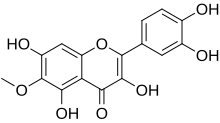
3,3′,4′,5,7-Pentahydroxy-6-methoxyflavone
Systematic IUPAC name
2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-6-methoxy-4H -1-benzopyran-4-one
Other names
6-Methoxyquercetin
Identifiers
ChEBI
ChEMBL
ChemSpider
ECHA InfoCard 100.007.529
EC Number
UNII
InChI=1S/C16H12O8/c1-23-16-9(19)5-10-11(13(16)21)12(20)14(22)15(24-10)6-2-3-7(17)8(18)4-6/h2-5,17-19,21-22H,1H3
N Key: JMIFIYIEXODVTO-UHFFFAOYSA-N
N InChI=1/C16H12O8/c1-23-16-9(19)5-10-11(13(16)21)12(20)14(22)15(24-10)6-2-3-7(17)8(18)4-6/h2-5,17-19,21-22H,1H3
Key: JMIFIYIEXODVTO-UHFFFAOYAR
COC1=C(C=C2C(=C1O)C(=O)C(=C(O2)C3=CC(=C(C=C3)O)O)O)O
Properties
C 16 H 12 O 8
Molar mass
−1
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
Patuletin is an O-methylated flavonol . It can be found in the genus Eriocaulon [1]
Glycosides Patuletin glycosides can be found in Ipomopsis aggregata [2]
Patuletin-3-O-rutinoside can be isolated from the aerial parts of Echinacea angustifolia [3]
Patuletin acetylrhamnosides can be isolated from Kalanchoe brasiliensis [4]
References
^ Bate-Smith, E. C.; Harborne, J. B. (1969). "Quercetagetin and patuletin in Eriocaulon". Phytochemistry . 8 (6): 1035. doi :10.1016/S0031-9422(00)86351-7 . ^ Smith, D. M.; Glennie, C. W.; Harborne, J. B. (1971). "Identification of eupalitin, eupatolitin and patuletin glycosides in Ipomopsis aggregata". Phytochemistry . 10 (12): 3115. doi :10.1016/S0031-9422(00)97361-8 . ^ Lin, L.; Qiu, S.; Lindenmaier, M.; He, X.; Featherstone, T.; Cordell, G. A. (2002). "Patuletin-3-O-Rutinoside from the Aerial Parts of Echinacea angustifolia". Pharmaceutical Biology . 40 (2): 92. doi :10.1076/phbi.40.2.92.5839 . S2CID 84855629 . ^ Costa, S. S.; Jossang, A.; Bodo, B.; Souza, M. L. M.; Moraes, V. L. G. (1994). "Patuletin Acetylrhamnosides from Kalanchoe brasiliensis as Inhibitors of Human Lymphocyte Proliferative Activity". Journal of Natural Products . 57 (11): 1503–1510. doi :10.1021/np50113a005 . PMID 7853000 .
Flavonols and their conjugates
Backbone
Flavonols
Aglycones Conjugates
Glycosides of herbacetin Glycosides of kaempferol
Afzelin (Kaempferol 3-rhamnoside)Astragalin (kaempferol 3-O-glucoside)Kaempferitrin (kaempferol 3,7-dirhamnoside)Juglanin (Kaempferol 3-O-arabinoside)Kaempferol 3-alpha-L-arabinopyranoside
Kaempferol 3-alpha-D-arabinopyranoside
Kaempferol 7-alpha-L-arabinoside
Kaempferol 7-O-glucoside Kaempferol 3-lathyroside
Kaempferol 4'-rhamnoside
Kaempferol 5-rhamnoside
Kaempferol 7-rhamnoside
Kaempferol 7-O-alpha-L-rhamnofuranoside
Kaempferol 3-xyloside
Kaempferol 7-xyloside
Robinin (kaempferol-3-O-robinoside-7-O-rhamnoside)Kaempferol 3-O-rutinoside Sophoraflavonoloside (Kaempferol 3-O-sophoroside)
Trifolin (Kaempferol 3-O-beta-D-galactoside) Glycosides of myricetin Conjugates of quercetin
O -Methylated flavonols
Aglycones Glycosides
of isorhamnetin
Narcissin (Isorhamnetin 3-O-rutinoside)
Isorhamnetin 3-O-glucoside
Tamarixetin 7-rutinoside other
Azalein (Azaleatin 3-O-α-L-rhamnoside)Centaurein (Centaureidin 7-O-glucoside)
Eupalin (Eupalitin 3-0-rhamnoside)Eupatolin (Eupatolitin 3-O-rhamnoside)Jacein (Jaceidin 7-O-glucoside)
Patulitrin (Patuletin 7-O-glucoside
Xanthorhamnin (Rhamnetin glycoside)
Derivative flavonols
Aglycones
Noricaritin
Dihydronoricaritin Glycosides
Pyranoflavonols
Furanoflavonols
Semisynthetic
The article is a derivative under the Creative Commons Attribution-ShareAlike License .
A link to the original article can be found here and attribution parties here
By using this site, you agree to the Terms of Use . Gpedia ® is a registered trademark of the Cyberajah Pty Ltd